News & Blogs 2023-09-18
Hepatitis B Virus Infection Worldwide

EN
CN
EN
News & Blogs 2023-09-18
Hepatitis B Virus Infection Worldwide
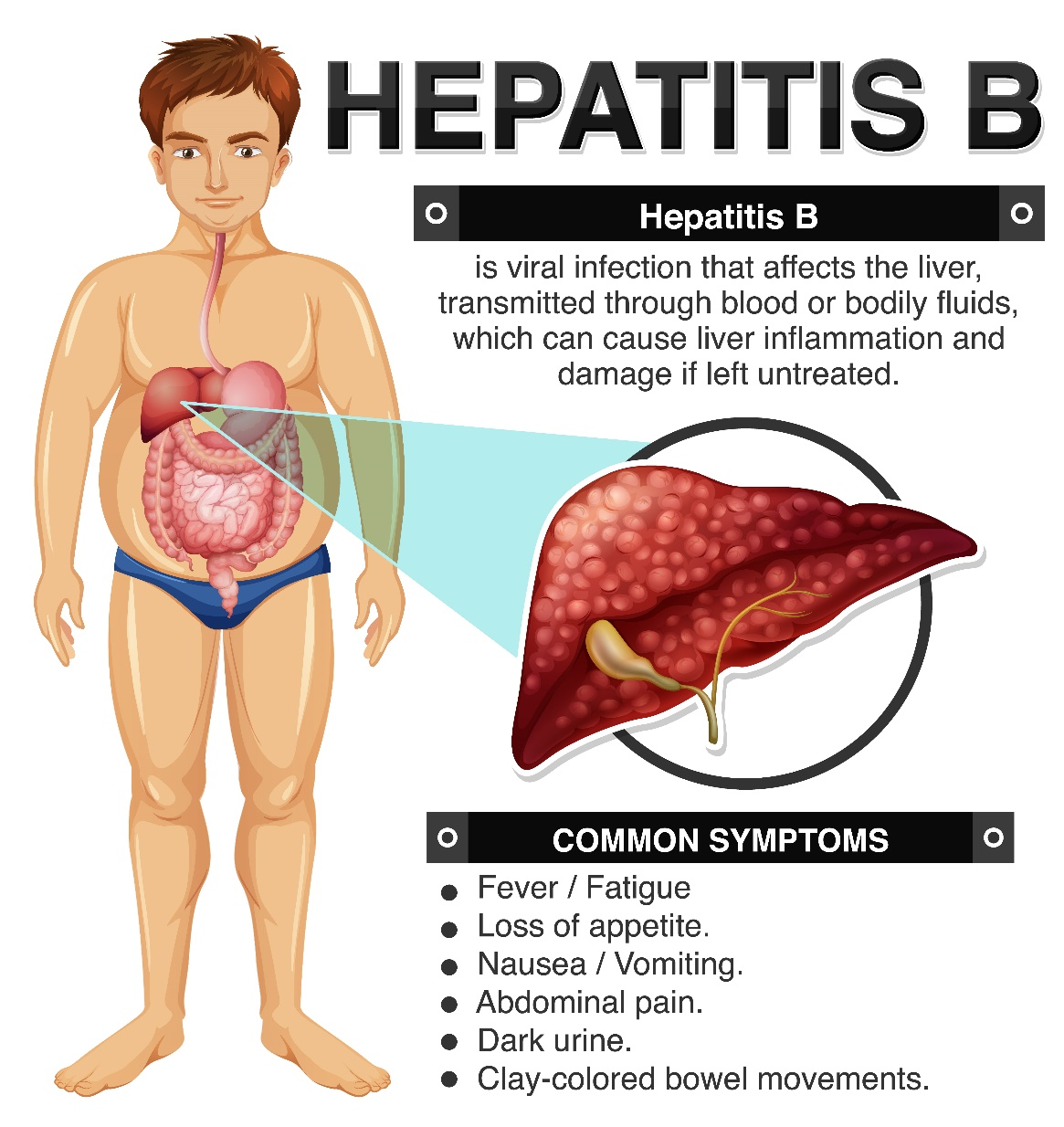
Hepatitis B virus (HBV) infection is a global health challenge of paramount concern. This minuscule yet formidable member of hepatophilic deoxyribonucleic acid (DNA) viruses has far-reaching implications across the world.
Upon infiltrating human liver cells, HBV embarks on a remarkable journey. A segment of its double-stranded circular DNA utilizes negative-stranded DNA as a template, meticulously repairing the cleaved region in the positive strand. The result is covalently closed circular DNA (cccDNA), a linchpin in the virus's replication cycle. From this cccDNA blueprint, a multitude of mRNA strands are transcribed, each encoding various antigens of the hepatitis B virus.
HBV's transmission vectors are diverse, including mother-to-child transmission, exposure to infected blood and blood products, contact with compromised skin mucous membranes, and sexual contact. The global impact is staggering. As of 2015, a staggering 257 million people were grappling with chronic HBV infection, with 887,000 succumbing to its effects.
While China has made significant strides in promoting the Hepatitis B vaccine, other countries grapple with the challenge. The urgency for enhanced surveillance, rapid case identification, and intervention is universal.
The prevalence of HBV infection exhibits significant regional variations. While some regions have made substantial progress in reducing HBV transmission through vaccination and healthcare initiatives, others continue to grapple with high infection rates.
High Prevalence Areas: Parts of sub-Saharan Africa and Southeast Asia bear a heavy burden of HBV infections, with chronic carriers accounting for a substantial portion of the population.
Moderate Prevalence Areas: Regions such as the Mediterranean, the Middle East, and Eastern Europe fall into the category of moderate HBV prevalence, with a notable but not overwhelming number of cases.
Low Prevalence Areas: Some countries in Western Europe, North America, and Australia have achieved low HBV prevalence rates thanks to successful vaccination programs and effective healthcare infrastructure.
HBV transmission occurs primarily through contact with infected blood or other body fluids. High-risk activities include unprotected sex, sharing needles for drug use, and perinatal transmission from mother to child during childbirth. Healthcare workers and individuals receiving medical or dental care in regions with inadequate infection control measures are also at risk.
The consequences of HBV infection are far-reaching. Chronic HBV infection can lead to severe liver diseases, including cirrhosis and liver cancer. The global mortality attributable to HBV is estimated at over 800,000 deaths per year. The economic and healthcare burdens of managing HBV-related diseases are substantial, particularly in areas with a high prevalence of the virus.
Despite these challenges, there is hope on the horizon. Vaccination campaigns have been instrumental in reducing HBV transmission in many countries. The World Health Organization (WHO) has set ambitious targets for the elimination of HBV as a public health threat by 2030, with a focus on expanding vaccination coverage, improving access to testing and treatment, and raising awareness about the virus.
As a fast-growing one-stop supplier in the global biopharmaceutical landscape, BIOEAST recognizes the universal need to combat HBV infection. Our cutting-edge raw material performance enables us to offer high-quality molecular and immunological raw materials, providing comprehensive solutions for the development of Hepatitis B virus diagnostic reagents worldwide.
At BIOEAST, we equip you with Nucleic Acid Extraction & Nucleic Acid Amplification products, arming you to join the global fight against hepatitis B virus infection. Together, we can make a difference in safeguarding the health of millions worldwide.
Nucleic Acid Extraction
Product Name | Cat No. | Sample Type |
Viral DNA Extraction Kit (Beads) | M1VD01 | Nasopharyngeal/oral swab, plasma, serum, tissue homogenate, cell culture fluid, cerebrospinal fluid, pleural fluid, ascites, aqueous humor, urine. |
Viral RNA/DNA Extraction Kit (Beads) | M1VR02 | Nasopharyngeal/oral swab, plasma, serum, tissue homogenate, cell culture fluid, cerebrospinal fluid, pleural fluid, ascites, aqueous. |
Nucleic Acid Amplification
Product Name | Cat No. | Introduction |
2× SYBR qPCR Master Mix | M4QS01 | qPCR premix with broad compatibility supports rapid amplification and high sensitivity. |
2× HotStart SYBR qPCR Master Mix | M4QS04 | Antibody-modified, highly sensitive qPCR premixes that support rapid amplification. |
2× Universal HotStart SYBR qPCR Master Mix | M4QS07 | Antibody-modified, highly sensitive qPCR premixes with broad compatibility to support rapid amplification. |
2× Taq Universal Probe Master Mix | M4QP01 | High assay specificity, high sensitivity, suitable for multiplexed qPCR assays. |
2× HS Taq Universal Probe Master Mix | M4QP02 | Antibody-modified, highly sensitive qPCR premixes with high assay specificity and sensitivity, suitable for multiplexed qPCR assays |
More products please refer to our molecular diagnostics page